Premium Introductory Organic Chemistry Course
.png)
Introductory Organic Chemistry Course covers the fundamentals of organic chemistry in 13 detailed chapters –
Atoms
Bonding in Atoms
.png)
Introductory Organic Chemistry Course covers the fundamentals of organic chemistry in 13 detailed chapters –
Atoms
Bonding in Atoms
The very premise of a covalent bond is electron sharing. As two atoms share one electron each to form one covalent bond, they may likely share more than once and form more bonds. This information on the number of connections between two atoms is revealed from the Bond Order.
So, the Bond Order measures the number of bonds between the two atoms in a molecule. The number can be integers like 1, 2, or 3 for single, double, or triple bonds or non-integers like 0.5, 1.3, 1.5, etc.
August Kekulé (1866) was the first to propose the alternating single and double bond structure of benzene that can only be possible if carbon could join other carbon atoms and form a ring.
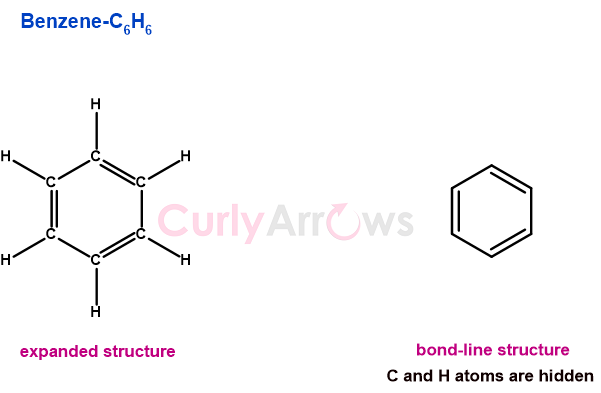
The first time the use of two dots or a colon (:) to represent two electrons acting as connecting links between the two atoms was proposed by G.N. Lewis in 1916.
In his landmark paper, ‘the atom and the molecules,’ he attempted to draw molecular structures using dots to represent the covalent nature of the chemical bonds, now popularly known as the Lewis structures.
So, a molecule of Cl2 was Cl : Cl, two electrons connecting two Cl atoms.
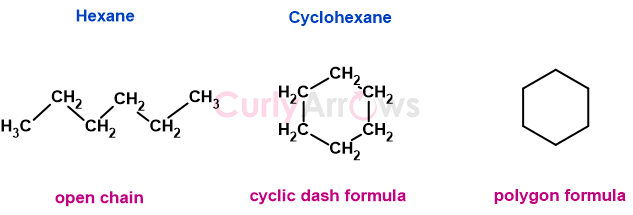
Organic compounds in which carbon atoms are joined in a ring, unlike the open-chain counterparts, are represented using the polygon formula. These cyclic polygons do not show carbon and hydrogen atoms. The corner of a polygon represents a carbon atom with its required number of hydrogen atom(s), and the sides of the polygon represent a carbon-carbon bond.
Molecular models are tools used for classroom or research to visualize better and interpret three-dimensional structures of organic molecules. The toolkit can be made of wood, plastic, metal, or other materials.
These models are digitally represented using computer graphics covered under molecular modelling, a branch of chemistry that uses computational and theoretical methods to model and mimic the molecule's behaviour in chemical and biological environments.
If a crystalline solid is cut with a sharp object, it would always give parts with smooth edges whereas an amorphous solid would cut into surfaces with rough, uneven edges. Crystalline solids are therefore said to have cleavage property, and amorphous solids do not show cleavage property.
Amorphous solids are said to be isotropic, and crystalline solids are anisotropic for their physical property measurements.
Isotropy comes from the Greek word; iso means same and tropos means direction. The name rightly indicates that for the amorphous solids; the physical property measurements are same in all the directions. The same correlation applies for anisotropy that means no same direction. It means that for the crystalline solids, the physical property measurements are not same in all the directions.

A matter can exist in four states- solid, liquid, gaseous, and plasma. The main characteristic that differentiates the solid from the other states of matter is its rigidity or hardness. The liquids and gases are not hard in nature and can flow; therefore, they possess fluidity.