In chemistry, intermolecular interactions are molecular interactions between neighbouring molecules, governed by attractive and repulsive forces.
Milk contains four major types of molecules: water, fats, carbohydrates, and proteins. These molecules look inert and motionless when you see a glass of milk, but their dynamic nature can be witnessed the moment you introduce a foreign agent, a few drops of dishwashing soap.
Materials You'll Need:
Milk (full-fat milk so that it is rich in fats and proteins)
Deep Dinner plate
Food colouring (few vibrant colours like red, yellow, green, or blue)
Dishwashing soap
Cotton swabs
Procedure:
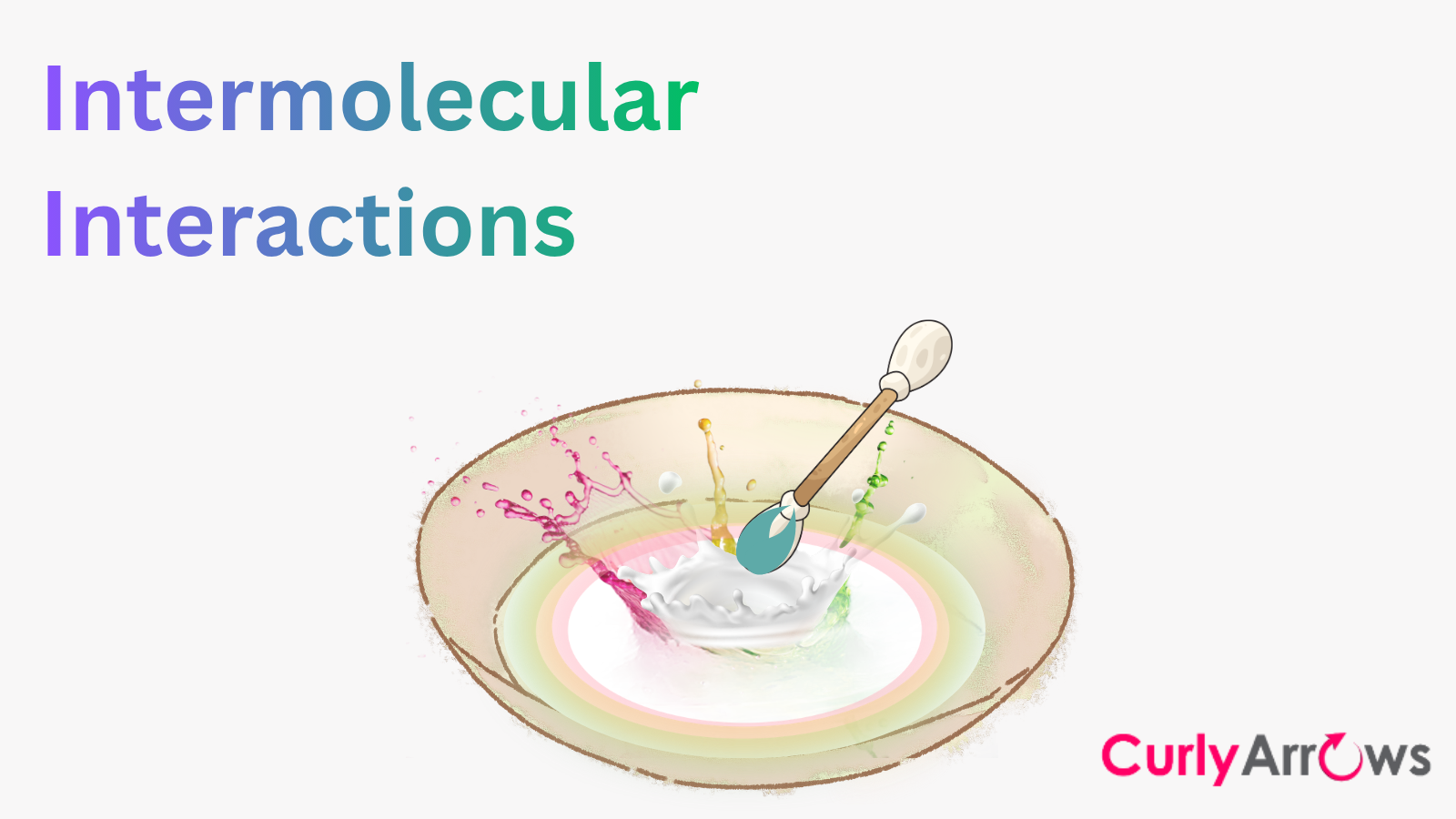
1. Pour enough milk into the deep dinner plate to completely cover the bottom to a depth of about 1/4 inch. Allow the milk to settle.
2. Add one drop of any vibrant food colour — red, yellow, blue, and green — in the centre of the plate of milk so that the drops fall close or on top of one another.
3. Take a clean cotton swab, dip one end into the centre of the milk without stirring it. Note the change.
4. Next, dip the other end of the cotton swab in a liquid dish soap, and this soapy end of the cotton swab should be dipped back in the middle of the milk. Hold for 10 to 15 seconds.
5. Taking another cotton swab, again dip it in liquid dish soap and place it at different corners in the milk.
Observation
Initially, when you touched a plain cotton swab in the centre of the milk containing food colours, nothing happened. However, when the swab containing dish soap was touched, the colours began to move, creating beautiful patterns no matter where you placed the dish soap-containing cotton swab.

Why It Works
Chemistry revolves around charges, positive and negative. These charges have an immense attitude. They don’t hang out with ‘like or similar’ charges, so remember the tag-line ‘like repels like’.
Dish soap also has two ends, positive and negative. Similarly, milk made of proteins, water, and carbohydrates has two opposite ends as well. Fat is mainly non-charge-containing molecules.

So, when the tip of the swab that contains a negative end interacts with the negative ends of the milk particles, the molecules repel and move away. Fat mainly acts as a middle child, a neutral peacemaker, butting itself in in these combative situations. The motion of colours across the plate, due to their extreme avoidance of molecules of similar types, is why milk becomes dynamic.
So, now we know that molecules are not literally visible in milk, but their intermolecular interactions, in this case repulsive, are observed in this colour-changing milk experiment. Molecules exist even when we don’t see it!
Related Content – Intermolecular forces (For Beginners, and Intermediates)
