Breadcrumb
- Home
- Organic Chemistry
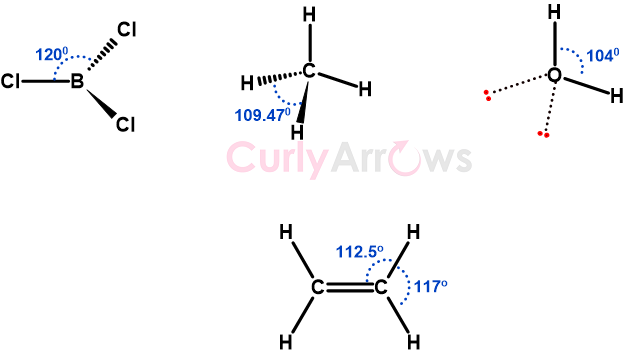
A bond angle is a geometrical angle between two bonds originating from the same central atom in a covalently bonded molecule, measured in degrees (o).
| Reactant | Reagent | Catalyst |
|---|---|---|---|
Definition | A substance that participates and undergoes a structural change in a chemical reaction is a reactant. | A reagent is a substance added to reactions intending to bring chemical changes. Sometimes it tests a reaction's progress and product formation(s). Therefore, it may or may not be consumed in a reaction. |
Bond lengths are smaller when atoms form multiple bonds. Single overlap between two atoms forms a single covalent bond. So, when they overlap twice and thrice to form double and triple bonds the atoms to come closer to form a tight-knit. Such bonds are even harder to break. So, the bond length of a triple is smaller than a double and single bond.
Carbon is naturally inclined to form new bonds, and in this quest, it may break away from the old ones. The atom or group of atoms that leaves the carbon chain to make way for the new bond is called the leaving group(s).
Such a bond dissociation can happen from a saturated, unsaturated, or carbonyl carbon.
Learning Objective: To learn about the structure, type, and nature of the carbanion intermediate part of organic chemistry reactions.
Skill Level - Intermediate
Prerequisites:
Nucleophiles are an atom or a group of atoms that are richer by two electrons and donate these electrons to electron-deficient species, the electrophiles.
Donating the electrons from the nucleophile to the electrophile creates a new two-electron covalent bond.
Electronegativity measures on a scale of 0.8 – 4 an atom’s or group of atoms’ tendency to attract the bond electron pair towards itself, thereby creating partial negative (δ-) and positive (δ+) terminals.
Groups in chemistry refer to the arrangement of elements of the periodic table into vertical columns. These vertical columns, called groups, are numbered from 1 to 18 and run from top to bottom. The first column on the left is labelled Group 1, and the last column on the right is labelled Group 18.
The elements in the group have the same number of electrons (valence electrons) in their outermost shell, also known as the valence shell, due to which they all have the same chemical or physical properties.