Breadcrumb
- Home
- Organic Chemistry
Organic chemistry and human beings both pursue betterment and advancement.
Humans engage in welfare activities to improve the well-being of others, tackle inequality, and create strong bonds. The same principle is also observed in organic chemistry.
Organic Chemistry stands out from other scientific disciplines due to its unique focus on carbon. This versatile element forms larger, more diverse molecules, and its reactions with other elements are crucial for enhancing reactivity, improving properties, introducing structural diversity, altering molecular composition, and more. These ambitious transformations are powered by electrons. Therefore, the study of organic chemistry begins with a deep understanding of its key players-carbon and electrons.
Keesom Forces | Debye Forces | London Forces |
|---|---|---|
The Keesom interactions occur in polar molecules with permanent dipoles. | Debye type of interactions occurs between the polar and nonpolar molecules. | Nonpolar molecules only interact by the London Forces.
|
a) Carbon and Silicon
b) Nitrogen and Phosphorus
c) Oxygen and sulfur
d) Magnesium and Calcium
Once the sigma bonds are formed, any additional electrons in the perpendicular unhybridized p-atomic orbitals engage in side-to-side or lateral overlap to form an additional bond known as the pi bond. The symbol π denotes the pi bond, drawn as an additional line over the sigma bond in the molecular structure.

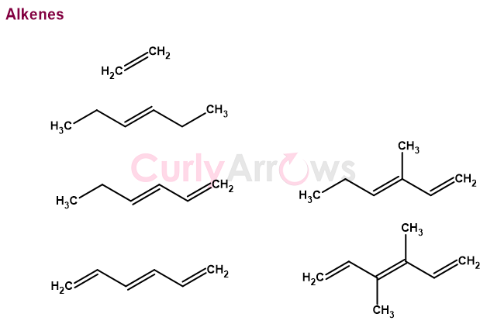
Alkene is a double bond containing hydrocarbon compound. Since the hydrocarbon compounds can be short or long chained, the double bond's position could be anywhere along the hydrocarbon chain.