Resonance in Chemistry
Pre-Requisite Reading: Lewis Structures, Types of reactions, Using curly arrows for electron movement, Identifying Functional groups.
Pre-Requisite Reading: Lewis Structures, Types of reactions, Using curly arrows for electron movement, Identifying Functional groups.
London dispersion forces are the weak intermolecular attractive forces that occur when the molecules are spaced closely. If the spacing is compact, the interactions are stronger. This means that the temporary dipoles induced due to the dispersion of electrons are also larger. Such a compact cluster will require more energy to break, raising the physical properties of the melting and boiling point.
The longest carbon chain is the parent chain containing the principal functional group.
The principle functional group is always given the highest priority, lowest number (1 before 2, 3, 4...), and assigned as a suffix while writing the nomenclature. Therefore, the numbering of the parent chain always starts from the principal functional group.
Structural isomerism is the variations in structures between two or more molecules having the same molecular formula.
Full Charges | Partial Charges | Transient Charges |
|---|---|---|
Atoms attain full charges (+ or -) by losing or gaining electrons and forming ions. So, a full charge on an ion is equivalent to the charge of a proton (in case of electron loss) or an electron (in case of electron gain).
|
Crystalline Solids | Amorphous solids | |
|---|---|---|
| Arrangement | The constituent particles are arranged in a regular order | The constituent particles show disordered arrangement |
| Order | Exhibit both long-range and short-range order | Exhibit only short-range order |
| Melting Point | Have sharp melting points | Solids melt over a range of temperatures |
Hydrogen bonding is a type of electrostatic interaction occurring in molecules in which the H atom is sandwiched between highly electronegative atoms like F, O, and N, one to which it is covalently bonded. For example, HF, H2O, NH3, etc.
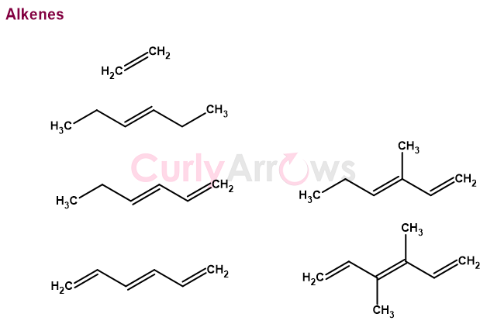
Alkene is a double bond containing hydrocarbon compound. Since the hydrocarbon compounds can be short or long chained, the double bond's position could be anywhere along the hydrocarbon chain.