Vander Waals Keesom (Dipole-Dipole) Interactions
Learning Objective: To study Vander Waals Keesom's force, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To study Vander Waals Keesom's force, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
Learning Objective: Study Vander Waals London forces, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
The atoms that form covalent bonds are the nonmetals (p-block elements), and they must have an electronegativity difference lower than 1.7. These values also help to classify the covalent bond into two kinds, polar and nonpolar.
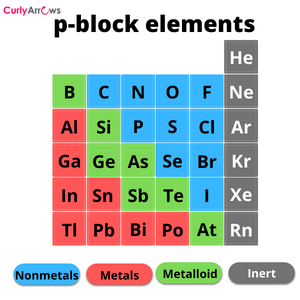
Once each electron of the two atoms forms the covalent bond, the electrons are simultaneously distributed between them so that they belong to both the atom’s nuclei. This area between the atoms is called the region of electron density.
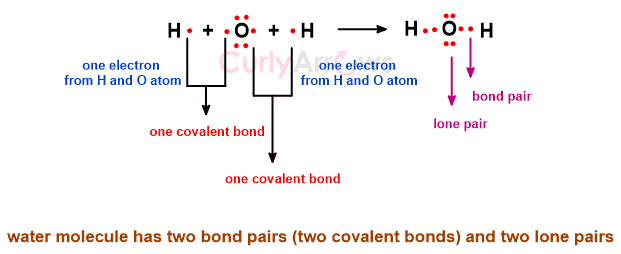
Once the outermost valence electrons of the atoms are engaged in the covalent bond formation to make a molecule, some electrons withhold themselves from the bond-making process. This divides the electrons in the molecule into two parts- bond pair and lone pair.
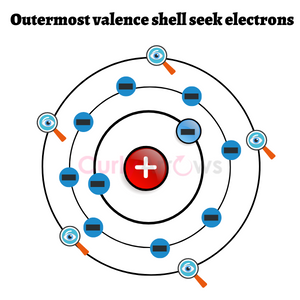
The atoms that embark on the journey of bond formation are likely to have an electron imbalance. They are less stable and become highly reactive. These atoms seek 8 electrons in their outermost shell to lower their reactivity and achieve stability.
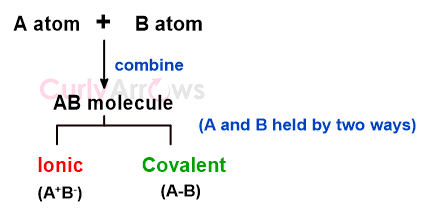
The Covalent bond is one of the two linkages by which atoms join to form molecules and is therefore classified under chemical bonds.
Pre-Requisite Reading: Lewis Structures, Types of reactions, Using curly arrows for electron movement, Identifying Functional groups.

Pre-Requisite Reading: Resonance
Solids are made up of many particles, and depending on how they are arranged, solids can be divided into crystalline solids and amorphous solids.
The particles may be atoms, ions, or molecules.
For example, gold atoms, NaCl ions, and sucrose molecules compose the solid-state.