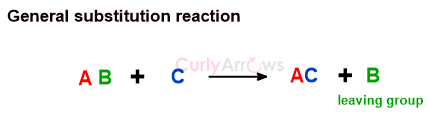
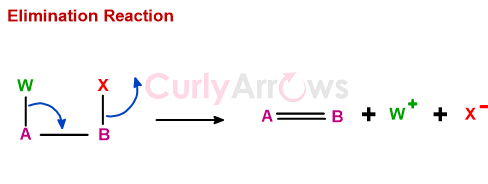
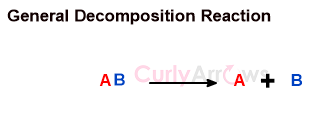
Oxidation-Reduction Type of Reactions in Organic Chemistry
Oxidation-Reduction Reaction in Organic versus Inorganic Chemistry
Inorganic chemistry describes oxidation in two ways. The first method is through the loss of electrons, and the second is by the increase in oxidation number.