Power of Protons - Structure of an Atom
Learning Objective: To learn about the components of an atom and the importance of proton numbers.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about the components of an atom and the importance of proton numbers.
Skill Level – Intermediate
Prerequisites:

Learning Objective: To study the types of hydrogen bonds, hydrogen bond strength, and significance with examples.
Skill Level – Intermediate
Prerequisites:
Solids are made up of many particles, and depending on how they are arranged, solids can be divided into crystalline solids and amorphous solids.
The particles may be atoms, ions, or molecules.
For example, gold atoms, NaCl ions, and sucrose molecules compose the solid-state.
Learning Objective: To learn about the trend and order of nucleophilicity for the p-block elements in the periodic table
Prerequisites:
Electronegativity trend of the elements from the Periodic table
When an atom is surrounded by six substituents, arranged in a manner that four are in one plane, one above and below, and their vertices join to give eight faces (octa-hedrons), such a molecular geometry is octahedral.
Since it looks like two pyramids projecting out from a square base, the geometry is also called square bipyramidal.
IUPAC, an international body engaged in developing standards for naming chemical elements and compounds, has laid several rules known as 'IUPAC rules' to identify a molecule by its parent alkane chain and the functional group. It has assigned various suffixes to identify organic chemistry's commonly seen functional groups.
London Dispersion, a type of Vander Waal Force, is the weakest of the three types, yet, it is the only one universally present in all the atoms and molecules.
In most cases, it is present in addition to the other forces; however, it is exclusively observed in nonpolar atoms and molecules devoid of any functional groups and, therefore, possess no other overpowering interactions. Examples are He, CH4, I2, C(CH3)4, etc.
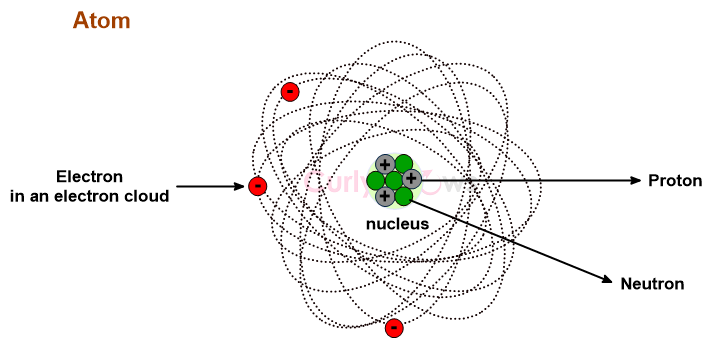
A Neutron is an uncharged, neutral subatomic particle found in an atom's nucleus, denoted as n or n0.
Branched alkyl chlorides (20 or 3o) cannot be used as a reagent in FC acylation reaction. The branched alkyl chlorides on forming acylium ion undergo the loss of CO to create a stable alkyl cation. The 20 or 3o alkyl cation reacts with benzene to form alkyl benzene.