Formation of Carbon Centered Free Radical
Learning Objective: To learn about different ways by which the formation of a carbon-centred free radical occurs.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about different ways by which the formation of a carbon-centred free radical occurs.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the importance of conformational analysis in organic chemistry.
Skill Level - Intermediate
Chapter: Stereochemistry
Sub-topic: Conformational Isomerism
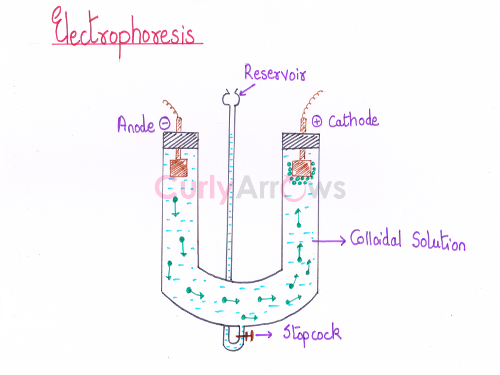
Depending on the nature of the charge (positive or negative) on the colloidal particles, its movement towards the electrode (cathode or anode) under the influence of an applied electric field is known as electrophoresis.
A branch of chemistry that deals with the interconversion of chemical energy and electrical energy taking place via the redox reactions.
It studies how a spontaneous redox reaction capable of generating chemical energy converts it into electrical energy. Example, Electrochemical cell.
Or how the use of electrical energy can bring about a non-spontaneous chemical reaction. Example, Electrolytic cell.
The bond angle is the angle at which two adjacent bonds converge and meet at the central atom in molecules.
Vander Waals is an attractive universal force that operates when the atoms and molecules are within a distance range of 0.4- 0.6 nm.
The attractive force closely gathers the atoms and molecules, so their collective strength can affect the state and other physical properties like melting and boiling points, viscosity, etc.
Aspect | Atomic Orbitals (AOs) | Molecular Orbitals (MOs) |
|---|---|---|
Definition | Simple regions around the nucleus of an atom where there’s a high probability of finding an electron and these regions is represented as spheres, or lobes. | A region in a molecule where electrons are likely to be found, formed by the combination of atomic orbitals of two or more atoms. |
Solution
The atoms or group of atoms are classified based on the Inductive effect as electron-withdrawing (-I) or electron-donating (+I) relative to Hydrogen.
The common functional groups showing +I and -I effect are:
+I group | -I group |
|---|---|
O- |