Introduction to Reactive Intermediates in Organic Chemistry
Learning Objective: To study what reactive intermediates are, their types, and their role in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study what reactive intermediates are, their types, and their role in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the structure, type, and nature of the carbanion intermediate part of organic chemistry reactions.
Skill Level - Intermediate
Prerequisites:
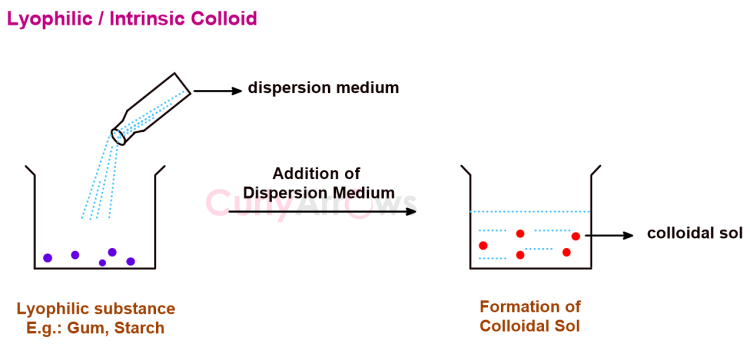
The substances that on mixing with a suitable dispersion medium readily form a colloidal sol are called lyophilic (solvent loving) and the sols thus formed are known as the lyophilic sols.
All atoms and molecules show London Dispersion Force, a primary way atoms and molecules interact. Most of the time, molecules have additional stronger forces than the London Dispersion Force, which is the weakest. Therefore, London Dispersion forces are exclusively seen in the nonpolar molecules since they don't have other competing functional groups leading to other interfering intermolecular interactions.
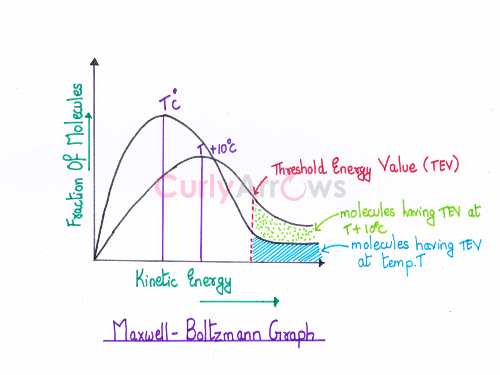
The peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy) corresponds to the most probable kinetic energy, and it is the energy possessed by the maximum fraction of the reactant molecules at a temperature T.
Benzyne | Benzyl | |
|---|---|---|
Identification | Benzyne is identified as a triple bond between two adjacent carbon atoms in a benzene ring. Root- Benzyne (like the triple bond of alkyne) |
The melting point of a substance is the temperature at which a solid, ordered crystalline substance is in equilibrium with a more random liquid state, thereby undergoing a phase change.
Since the components of the solid substances are bound by strong intermolecular attractive forces, it also reflects the energy needed to weaken these attractive forces and move freely in the liquid state.
Keesom Forces | Debye Forces | London Forces |
|---|---|---|
The Keesom interactions occur in polar molecules with permanent dipoles. | Debye type of interactions occurs between the polar and nonpolar molecules. | Nonpolar molecules only interact by the London Forces.
|
The very premise of a covalent bond is electron sharing. As two atoms share one electron each to form one covalent bond, they may likely share more than once and form more bonds. This information on the number of connections between two atoms is revealed from the Bond Order.
So, the Bond Order measures the number of bonds between the two atoms in a molecule. The number can be integers like 1, 2, or 3 for single, double, or triple bonds or non-integers like 0.5, 1.3, 1.5, etc.
Learning Objective: To study the three types of addition reactions that nucleophiles undergo.
Skill Level - Intermediate
Prerequisites: