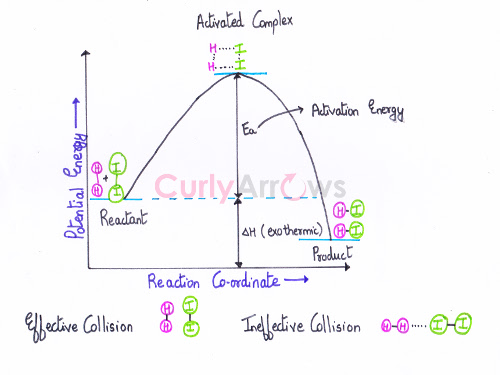
Effective Collisions
Effective collisions result in product formation due to an increase in the rate of a chemical reaction. It occurs when the two reactant molecules are correctly oriented and have attained the threshold value (or the activation energy value) at the time of the collision.