Atomic Orbitals - S, P, D, F
Learning Objective: To learn about the different types of orbitals housing electrons in an atom, their visual representation, meaning, and their energy levels.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about the different types of orbitals housing electrons in an atom, their visual representation, meaning, and their energy levels.
Skill Level – Intermediate
Prerequisites:

| Electrofuge | Electrophile |
|---|---|---|
Definition | Electrofuge is a leaving group that is formed due to the heterolytic breakage of a bond wherein post the clevage, it leaves without the bond pair of electrons, and therefore electron deficient. | Electrophiles are electron-deficient species that may be neutral or charged because of heterolytic bond cleavage. Still, its primary nature is to attract electrons from other electron-rich counterparts and form a new bond. |
Water at room temperature is liquid in nature due to the hydrogen bonds holding the water molecules (H2O). The close association is hard to break; therefore, water boils at a high temperature of 100oC. The high temperature breaks the Hydrogen bond links to free individual H2O molecules.
London dispersion forces are the weakest intermolecular forces among all the van der Waal forces of interactions. This interaction occurs in molecules due to electrons' continuous random motion, creating a temporary dipole. The instantaneous dipole in one molecule induces a dipole in the others, causing dispersion. The molecules then start to show intermolecular attraction.
When the atoms combining to form molecules are of the same type, it is a homoatomic molecule. For example, when two Hydrogen atoms (2H) combine under an appropriate reaction condition, a Hydrogen molecule (H2) is formed.
A homoatomic molecule can be diatomic (like elemental hydrogen, H2), triatomic (like ozone, O3), or polyatomic (like sulphur allotrope, S8).
Learning Objective: To study the various examples of organic reactions where the leaving group departs from a molecule and also understand its various types.
Prerequisites:
Displacement and Elimination type of reaction (Chapter- Common Types of Reactions)
Atoms generally form bonding arrangements that give them filled shells of electrons like a noble gas configuration. The stability the atom aims for is that of its nearest noble gas.
For example, Lithium (Z = 3, Electronic Configuration = 1s2, 2s1) of the second row would prefer losing one electron to become Li+ (Z = 3, Electronic Configuration = 1s2) having an electronic arrangement similar to its nearest noble gas Helium (Z = 2, Electronic Configuration = 1s2).
A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
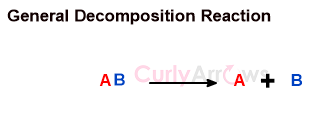
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.