Breadcrumb
- Home
- Organic Chemistry
Free radicals are generated due to the homolytic bond cleavage, a type of bond breaking where each atom holding the two-electron covalent bond gets one electron. Such a homolytic bond cleavage requires an input of energy, either in the form of heat or light.

Solution

A sp3 hybridized carbon is a tetravalent carbon that forms four single covalent bonds with itself or atoms of other p-block elements to its right, namely Oxygen, Carbon, Nitrogen, and Halogens. It also forms a bond with elements capable of forming covalent bonds, such as hydrogen.
The bonds formed are of equal strength and at an angle of 109.5o due to which the central carbon atom is tetrahedral in shape. Example, carbon of an alkane or an alkyl group.
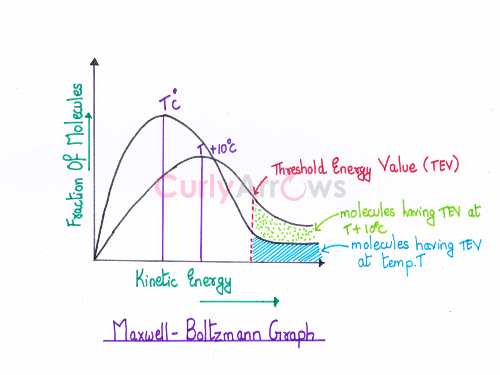
The peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy) corresponds to the most probable kinetic energy, and it is the energy possessed by the maximum fraction of the reactant molecules at a temperature T.
The condensed structural formula provides the shortest way to understand the atoms and their numbers, their connection with respect to each other, and therefore, visualize the compounds' structure without elaborately drawing it, but by writing it in a single line.
So, if the elaborate structure of butanoic acid look like A, the condensed structural formula would look like B.
Benzene | Benzyne | |
|---|---|---|
Identification | Alternating single and double bonds. | Alternating single and double bonds plus an extra pie bond (triple bond) between two adjacent carbons. |
Structure |