Bonding in Atoms
Atoms primarily form three types of bonds – ionic, covalent, and metallic.
We start by exploring the Octet Rule that determines Ionic and Covalent bonds.

Atoms primarily form three types of bonds – ionic, covalent, and metallic.
We start by exploring the Octet Rule that determines Ionic and Covalent bonds.

Intermolecular forces are responsible for holding various molecules and ions close to each other. Their attractive strength is significant enough to affect various physical, chemical, and biological properties.
Most probable kinetic energy is the energy possessed by the maximum number of reactant molecules at a temperature T, and it is shown as a peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy).
| Types of Interactions | Shown By | Energy (KJ/mole) | Examples (Image) |
|---|---|---|---|
| Ion-Ion | Only Ions (E.g.- Na+Cl- or carboxylate ion and ammonium ion) | 400-4000 |  |
| Ion-Dipole |
Learning Objective: To investigate which intermolecular forces are predominant in a liquid state and to study the impact of molecular weight, molecular shape, and polarity on the boiling point of liquids.
Skill Level - Intermediate
Prerequisites:
Metallic Bond | Ionic Bond |
|---|---|
| Metals have metallic bonding in them. Ex: Na, K. | Metals and nonmetals participate in forming the Ionic bond. Ex: NaCl, KBr. |
Several metal atoms lose valence electrons and become positive kernels holding a fixed lattice position. The lost electrons then freely float between the positive kernels to make the structure electrically neutral. |
A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
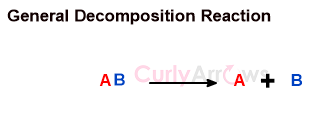
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.
Sharing resources is essential to build a harmonious world. When the resources are shared ineffectively conflicts emerges. A similar principle extends to Chemistry.
Atoms are in a state of harmony when the neighbouring atoms shares electrons. When the sharing stops, Chemical reactions triggers.
The imbalance in sharing electrons influences the molecule’s polarity, reactivity, and physical properties. The disagreement on sharing is brought about by electronegativity.
In the electrochemical series, the electrodes are arranged in the increasing order of their reduction potential under standard conditions of 1M electrolyte concentration, 298 K temperature, and 1 bar atmospheric pressure.
It is also known as the activity series as the activities of two different electrodes towards displacement reaction are compared; whether the electrode under standard reaction condition will have reduction potential or oxidation potential.
Learning Objective: To study the three types of addition reactions that nucleophiles undergo.
Skill Level - Intermediate
Prerequisites: