What are the properties of metallic bonds?
Several characteristic properties of the metals are due to the nature of the metallic bond. Some unique properties include-
1. High Melting and Boiling points:
Several characteristic properties of the metals are due to the nature of the metallic bond. Some unique properties include-
1. High Melting and Boiling points:
- The unit cells that makes up the entire crystalline solid has a fixed value for the length of the edges and the angle between the edges.

- Therefore, the heat energy required to melt a crystalline solid would have a fixed value.
London dispersion forces are the weak intermolecular forces that occur when the molecules are in close proximity to each other. The strength of these forces is determined by the atomic size of the element.
In smaller atoms, the atomic radius is smaller, which means that the positively charged nucleus holds the outermost valence electrons more firmly, leaving less room for their dispersion or polarization.
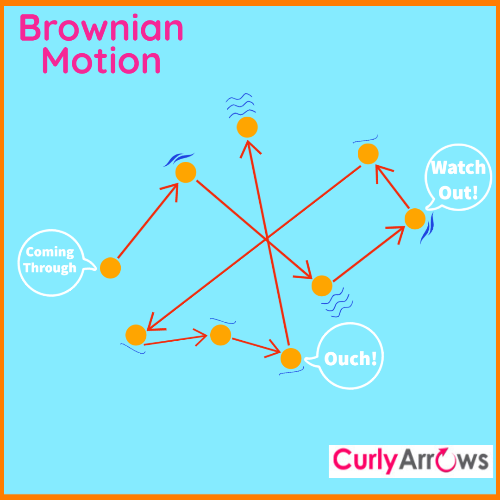
On a fine morning in 1827, botanist Robert Brown had no idea that a simple observation through his microscope would lead to the discovery of invisible matter.
The strength of polar-nonpolar interactions is affected by the atomic size of the nonpolar atom (or molecule) and the electronegativity of the polar molecule.
Debye intermolecular forces of attraction occur between polar and nonpolar atoms or molecules. When closely placed with a polar molecule (electronegativity difference between 0.5-1.7), the electron cloud of the nonpolar atom (or molecule) is influenced and distorted, leading to the formation of temporary dipoles. As the polarity is induced (forced) in the otherwise nonpolar atom, the Debye forces are said to show an Induction effect.
Metallic solid consists of several metal atoms of the same kind bonded together closely. Due to their large size, metals easily lose their outermost valence electrons. The delocalized valence electron drifts and is now available for the other nuclei of metal atoms. Several delocalized electrons are comparable to a sea of electrons.
A molecule is said to be polar if the bond electrons between the atoms in a covalent bond are unequally distributed, creating two ends. The electron redistribution is marked by showing the separation of charges as a partial charge above the atom's symbol. Therefore, a polar molecule always has a positive and a negative end resulting in a dipole. The pulling of bond electrons by an atom is attributed to its electronegativity.
Keesom forces occur in polar molecules where the atoms have a considerable electronegativity difference, resulting in the charge separation and formation of positive and negative poles. The negative pole of one molecule electrostatically interacts with the positive pole of the neighboring molecule. Consequently, all the polar molecules in the medium try to orient their dipoles to be in alignment for such electrostatic interactions.
Bi (V) represents the +5-oxidation state of Bismuth (Bi). It means Bi has lost 5 electrons from its valence shell. Bi, therefore, wants to gain the lost electrons and is called an oxidizing agent. The process of electron uptake is a reduction reaction.
Bi (V) is a stronger oxidizing agent than Sb (V) and, therefore, will pick up the electrons faster than Sb (V). Due to the electron uptake, it will then go from a +5-oxidation state to a +3-oxidation state. Bi is now less by only three electrons due to the two-electron gain.