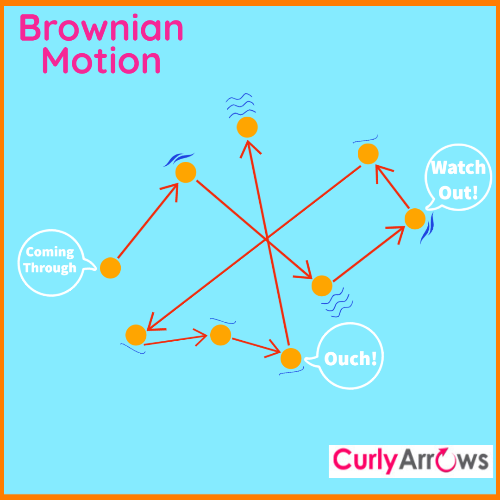
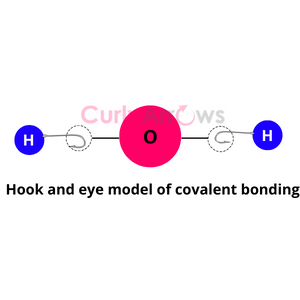
What is known as the Most Probable kinetic energy? (Collision Theory Chemistry)
Most probable kinetic energy is the energy possessed by the maximum number of reactant molecules at a temperature T, and it is shown as a peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy).