Breadcrumb
- Home
- Organic Chemistry
Russian chemist Vladimir Markovnikov proposed Markovnikov’s rule in 1869 to predict the regiochemistry of addition reactions between unsymmetrical alkenes/ alkynes and hydrogen halides to form alkyl halides.
According to the rule, ‘An alkyl halide is formed in a reaction between an unsymmetrical alkene or alkyne and hydrogen halide (HX) when the negative part of the reagent (X-) attaches to the alkene carbon that has fewer number of hydrogen atoms across the double bond.’
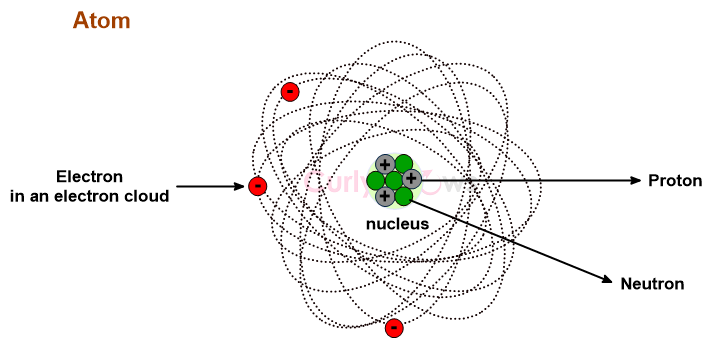
A Neutron is an uncharged, neutral subatomic particle found in an atom's nucleus, denoted as n or n0.
| sp3 | sp2 | sp |
|---|---|---|---|
Orbitals involved | One s and three p-orbitals of the central atom that are close in energy mix to form four sp3 hybrid orbitals for covalent bond formation. |
Inductive Effect | Resonance/Mesomeric Effect |
|---|---|
The sigma (σ) or the covalent bond electrons are pulled or pushed so that the electron density shifts towards the most electronegative atom. | The pie (π) electrons are pulled and pushed through the p-orbitals along the sigma (covalent) bond framework. |
It is a permanent effect. | It is a permanent effect. |
Learning Objective: To learn through an example how a reaction outcome depends on the conformation of the reactant.
Skill Level - Intermediate
Prerequisites:
Ionic Bond | Covalent Bond | Metallic Bond | Van der Waal Forces |
|---|---|---|---|
Ionic bonds form due to the complete transfer of electrons. | Covalent bonds form due to sharing of electrons. | Metallic Bond forms between a Metal (cation) and delocalized electrons. |
The crystalline solid is made up of the unit cells. Therefore, the properties of the unit cell turn out to be the properties of the crystalline solid. In a unit cell, the length of the edges and the angle between the edges are fixed. Therefore, the entire crystalline solid would exhibit fixed value for both these parameters.