Ion
Ion is different from an atom since an atom is an electrically neutral specie with an equal number of positive protons and negative electrons.
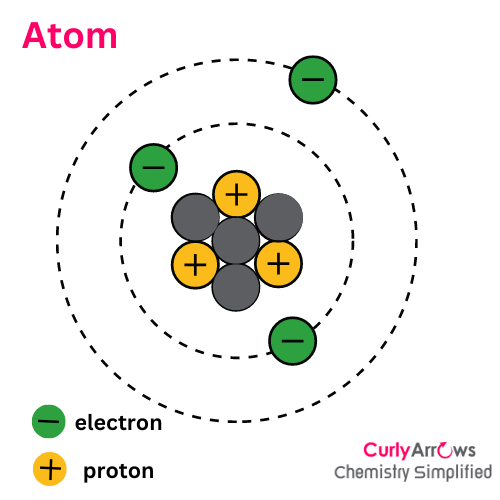
Ion is different from an atom since an atom is an electrically neutral specie with an equal number of positive protons and negative electrons.


Dalton, in 1804, in his work on ‘Atomic Theory,’ proposed the law of chemical combination to explain how atoms form compounds. According to him, atoms of different elements combine in a simple whole-number ratio to give compounds.
For example, two Hydrogen and one Oxygen combine to form water (H2O), or four Hydrogens and one Carbon combine to form methane (CH4), or one Nitrogen combines with three Hydrogens to give ammonia (NH3).
Learning Objective: To learn about the factors affecting ion-dipole interaction strength.
Skill Level - Intermediate
Prerequisites:
Valence Bond (VB) Theory | Valence Shell Electron Pair Repulsion (VSEPR) Theory |
|---|---|
Valence Bond Theory explains how atoms combine to form di and polyatomic molecules held by covalent bonds. | VSEPR theory considers shape, molecular geometry, and bond angles as an after-effect of covalent bond formation. The molecules take up various shapes to overcome the electron repulsions between the bonding and nonbonding electrons of the combining atoms. |

Identifying a change is the first step in solving or designing a chemical reaction.
Solution
| Condensed Formula | Molecular Formula |
|---|---|---|
Definition | The condensed formula is a shorthand representation of a molecule that gives information on the atoms, their number, and how they are connected in a single line.
| The molecular formula represents the total number of atoms in a compound without showing any connection between the atoms.
|
The regular, periodic arrangement of atoms, ions or molecules at the lattice points of a crystal in a three-dimensional space is called the space lattice or the crystal lattice.
