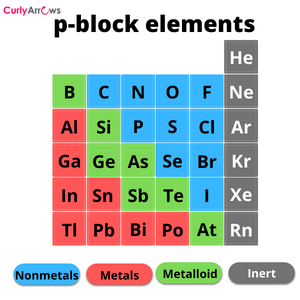
Groups
Groups in chemistry refer to the arrangement of elements of the periodic table into vertical columns. These vertical columns, called groups, are numbered from 1 to 18 and run from top to bottom. The first column on the left is labelled Group 1, and the last column on the right is labelled Group 18.
The elements in the group have the same number of electrons (valence electrons) in their outermost shell, also known as the valence shell, due to which they all have the same chemical or physical properties.
.png)