Isomers are molecules with the same molecular formula; that is, they have the same atoms in exact numbers (or the same molar masses); however, they still differ in their structures.
These differences or structural variations arise due to the atom’s attachment styles.
An atom may connect differently, each time having different set of neighbors by bond or in space.
This change in arrangement affects their overall chemical and physical properties. Therefore, though they have a common molecular formula, they are still unique.
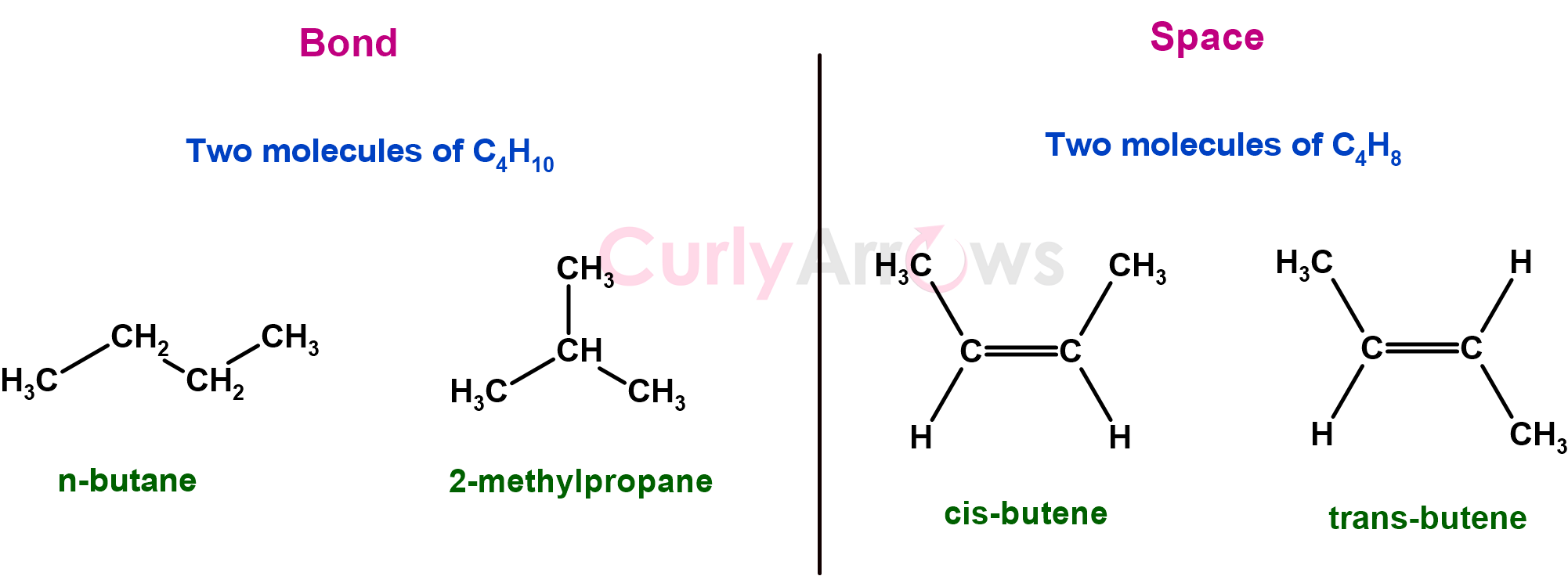
For example, n-butane and 2-methylpropane have the same molecular formula (C4H10) but are two completely different molecules by bond. If n-butane’s boiling point is -1oC, it is -11.6 oC for 2-methylpropane.
Related Reading: How change in structure affects the physical properties of organic compounds (subscribers only premium tutorial)
On the other hand, cis-butene and trans-butene are similar in how the atoms are arranged in bonds. However, they differ in the spatial arrangement of the methyl (-CH3) groups that are placed differently across the double bond. Cis-butene reacts differently from trans-butene when reacted with HBr in ether, indicating a difference in their chemical properties.
Related reading: Markovnikov’s rule of alkene addition (free)
Stability of substituted alkenes (free)
Such molecules that exist as isomers are said to show isomerism. In simpler words, isomerism is the phenomenon that suggests the possibility of the existence of isomers.
Carbon has a remarkable ability to be tetravalent due to which it can form long or branched chains, contain multiple bonds on the chain, or even contain heteroatoms and diverse functional groups. In addition, such a tetravalent carbon can even hold four different groups on one atom, making such a carbon center chiral. Due to all these properties of carbon compounds, they are known for exhibiting different forms of isomerism in organic chemistry.
Organic chemistry broadly categorizes isomerism into two types-
- Structural isomerism is also known as constitutional isomers (bonds between the atoms differ).
Stereoisomerism is also known as spatial isomers (relative positions of the atoms differ).
Related Chapter: Structural Isomerism in Organic Chemistry (subscribers only premium tutorial)
