Molecular Orbital
When atoms engage to form molecules, they do so by the overlap of atomic orbitals, creating a bigger molecular orbital that now encompasses more than one atom of a molecule.
When atoms engage to form molecules, they do so by the overlap of atomic orbitals, creating a bigger molecular orbital that now encompasses more than one atom of a molecule.
A molecule is an electrically neutral chemical entity that consists of two or more bond atoms.
The joining of two or more atoms by a linkage known as a covalent bond creates molecules such that changing the connection, or the arrangement will change the molecule and its properties.
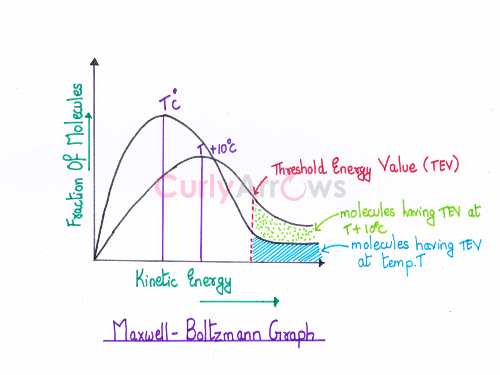
The peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy) corresponds to the most probable kinetic energy, and it is the energy possessed by the maximum fraction of the reactant molecules at a temperature T.
On the dissolution of a substance in a dispersion medium, the aggregation of a vast number of atoms or smaller molecules of varying sizes (< 10-9 m) leads to the formation of colloidal particles of the size range 10-9 to 10-6 m.
As the colloidal particles are now made up of many atoms or molecules, it is now called multimolecular colloids.
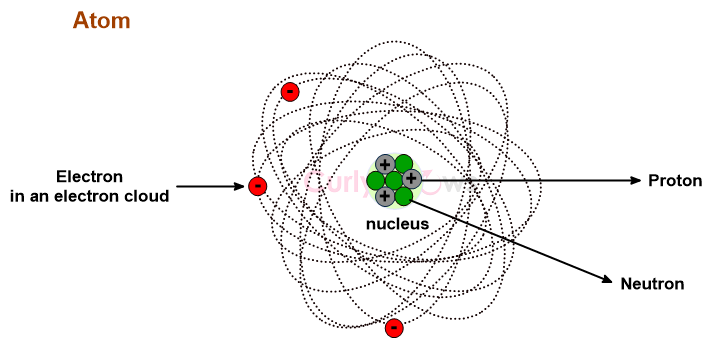
A Neutron is an uncharged, neutral subatomic particle found in an atom's nucleus, denoted as n or n0.
Newman Projection is a way of representing the spatial relationship of groups attached to two adjacent carbon atoms in a molecule. The angle at which the observer makes this observation occurs when they try to visualize the molecule head-on along the bond axis joining the two atoms, such that one carbon atom is seen as a front and the other as a rear.
Nucleophiles are an atom or a group of atoms that are richer by two electrons and donate these electrons to electron-deficient species, the electrophiles.
Donating the electrons from the nucleophile to the electrophile creates a new two-electron covalent bond.
An atom has a central, dense, tightly packed nucleus with a diameter of 10-15 m compared to an atom at 10-10 m, a size equivalent to a marble in a football stadium. Therefore, the nucleus makes up less than 0.01 % of the atom's volume.
The nucleus contains positively charged protons and neutral neutrons as subatomic particles bound firmly by the nuclear force.
When an atom is surrounded by six substituents, arranged in a manner that four are in one plane, one above and below, and their vertices join to give eight faces (octa-hedrons), such a molecular geometry is octahedral.
Since it looks like two pyramids projecting out from a square base, the geometry is also called square bipyramidal.
Atoms generally form bonding arrangements that give them filled shells of electrons like a noble gas configuration. The stability the atom aims for is that of its nearest noble gas.
For example, Lithium (Z = 3, Electronic Configuration = 1s2, 2s1) of the second row would prefer losing one electron to become Li+ (Z = 3, Electronic Configuration = 1s2) having an electronic arrangement similar to its nearest noble gas Helium (Z = 2, Electronic Configuration = 1s2).