Electrophoresis
Depending on the nature of the charge (positive or negative) on the colloidal particles, its movement towards the electrode (cathode or anode) under the influence of an applied electric field is known as electrophoresis.
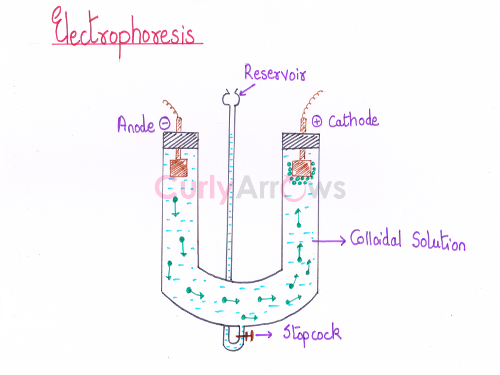
Depending on the nature of the charge (positive or negative) on the colloidal particles, its movement towards the electrode (cathode or anode) under the influence of an applied electric field is known as electrophoresis.

A type of electrophilic aromatic substitution reaction to introduce an acyl group (R-C=O) in the aromatic ring resulting in aryl ketones (Ar-COR). The reacting species are usually but not always -benzene (substituted or unsubstituted), an acyl halide and a Lewis Acid catalyst like AlCl3.
A type of electrophilic aromatic substitution reaction that introduces an alkyl group (-R) in the arene ring, giving alkyl arenes.
The reacting species are usually but not always- benzene ring (substituted or unsubstituted), an alkyl halide and a Lewis Acid catalyst like AlCl3.
Groups in chemistry refer to the arrangement of elements of the periodic table into vertical columns. These vertical columns, called groups, are numbered from 1 to 18 and run from top to bottom. The first column on the left is labelled Group 1, and the last column on the right is labelled Group 18.
The elements in the group have the same number of electrons (valence electrons) in their outermost shell, also known as the valence shell, due to which they all have the same chemical or physical properties.
When atoms of different types combine to form molecules, it is a heteroatomic molecule. For example, when Carbon (C) and Oxygen (O) atoms combine under an appropriate reaction condition, it can lead to the formation of two types of products (carbon monoxide, CO, and carbon dioxide, CO2) containing two types of atoms.
A heteroatomic molecule can be diatomic (like hydrogen chloride, HCl), triatomic (like water, H2O), or polyatomic (like methane, CH4).
Heterolytic cleavage or heterolysis is a chemical reaction in which the bond between two atoms breaks unequally so that the two bond electrons reside with only one atom.
The atom that receives both electrons become an electron-rich negatively charged ion (anion, denoted with a negative sign), and the atom that lost the electrons forms a positively charged ion (cation, denoted with a positive sign).
When the atoms combining to form molecules are of the same type, it is a homoatomic molecule. For example, when two Hydrogen atoms (2H) combine under an appropriate reaction condition, a Hydrogen molecule (H2) is formed.
A homoatomic molecule can be diatomic (like elemental hydrogen, H2), triatomic (like ozone, O3), or polyatomic (like sulphur allotrope, S8).
Homolytic cleavage, or homolysis, is a chemical reaction in which a covalent bond between two atoms is broken equally, and each atom retains one of the two electrons that form the bond. This creates two species (similar or dissimilar), each with an unpaired electron, known as the radicals. The radical electron is denoted with a dot (.) over the atoms’ symbol.
Hydrogen bonding is an attractive intermolecular interaction stronger than Vander Waals forces that exclusively occurs when the Hydrogen atom is sandwiched between two highly electronegative atoms N, O, or F, one to which it is covalently bonded and the other it attracts electrostatically.
An atom or group of atoms that can pull the bond electrons towards itself or push the bond electrons from itself and decreasingly transmit the effect along the sigma (σ) bonds of the carbon chain inducing permanent polarization in the molecule. Such an effect is called the Inductive effect.