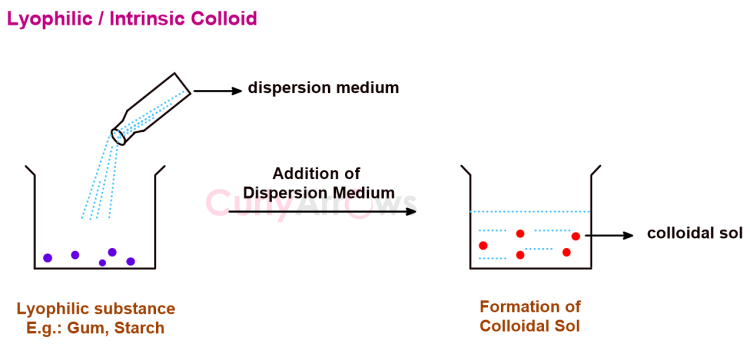
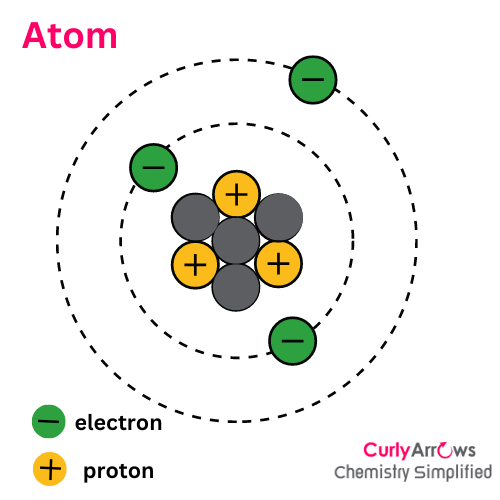
Intermolecular Forces
Intermolecular forces are collective forces in organic chemistry that cause atoms and molecules to stick together and interact; therefore, they are electrostatic in nature. The collective strength of these electrostatic interactions can affect a compound's physical properties, like melting and boiling points, density, refractive index, solubility, and others.
Almost all atoms and molecules interact through intermolecular forces, an inherent property resulting from the presence of electrons.