Bond Angle
A bond angle is a geometrical angle between two bonds originating from the same central atom in a covalently bonded molecule, measured in degrees (o).
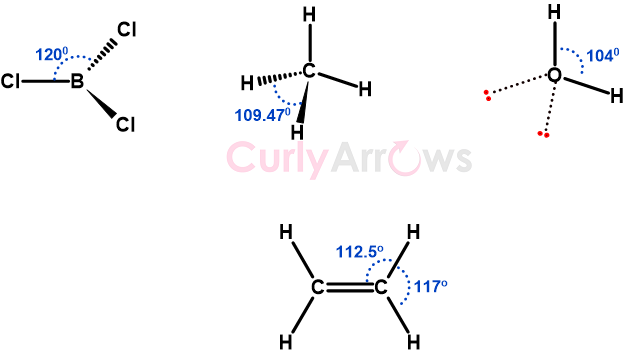
A bond angle is a geometrical angle between two bonds originating from the same central atom in a covalently bonded molecule, measured in degrees (o).

The random collisions of the colloidal particles (dispersed phase) with molecules of the dispersion medium (colloidal sol) that results in a zig-zag movement of the colloidal particles, when observed under an ultramicroscope, is known as the Brownian Motion.
A carbanion is a negatively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions.
With three bonds and 8 electrons, carbanions are electron rich and act as a base forming a C-H bond or as a nucleophile forming a C-C bond.
A carbocation is a positively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions.
With three bonds and only six electrons, carbocations have an incomplete octet and, therefore, electron deficient. It functions as an electron acceptor and an electrophile forming new Carbon-Carbon (C-C) bonds.
In molecules consisting of more than two atoms, the least electronegative atom (except Hydrogen) is the central atom. Due to its low electronegativity, the central atom will not hoard electrons but will share with other atoms, thereby forming a maximum number of bonds than the terminal atoms. So, the central atom is also the least numerous.
Collision Frequency in chemical kinetics is defined as the number of collisions that take place per second per unit volume of the reaction mixture between two reactant molecules considered as hard spheres. It is represented as Z.
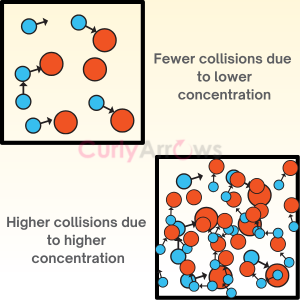
Collision theory is applicable only to bimolecular reactions mostly gases where the reactant molecules are considered as hard spheres that must collide with sufficient threshold energy and must be correctly oriented for the collision to be effective that results in the product formation.
The rate of the reaction, therefore, depends on the collision frequency, threshold energy, and the orientation (steric/probability) factor.
k= PZABe-Ea/RT
where,
The different spatial arrangements organic molecules adopt due to the rotation of the single bond are called conformations. A specific conformation is called a conformer or conformational isomer.
Many such conformational isomers are undoubtedly interconvertible by single bond rotations. So, these isomers are simply different rotational (or structural) arrangements of the same molecule.
The compounds in which the central metal atom is linked to ligands (anions or neutral molecules) that donate its pair of electrons to form coordinate covalent bonds with the metal atom.
Example, Ni(CO)4, {Co(NH3)6]Cl3
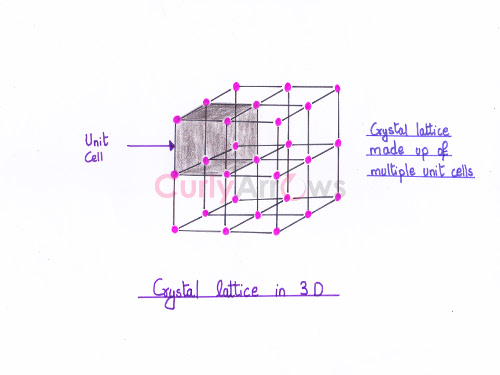
The regular, periodic arrangement of atoms, ions or molecules at the lattice points of a crystal in a three-dimensional space is called the space lattice or the crystal lattice.